
- what element has 7 protons 7 neutrons and 10 electrons
- what element has 7 protons 8 neutrons and 10 electrons

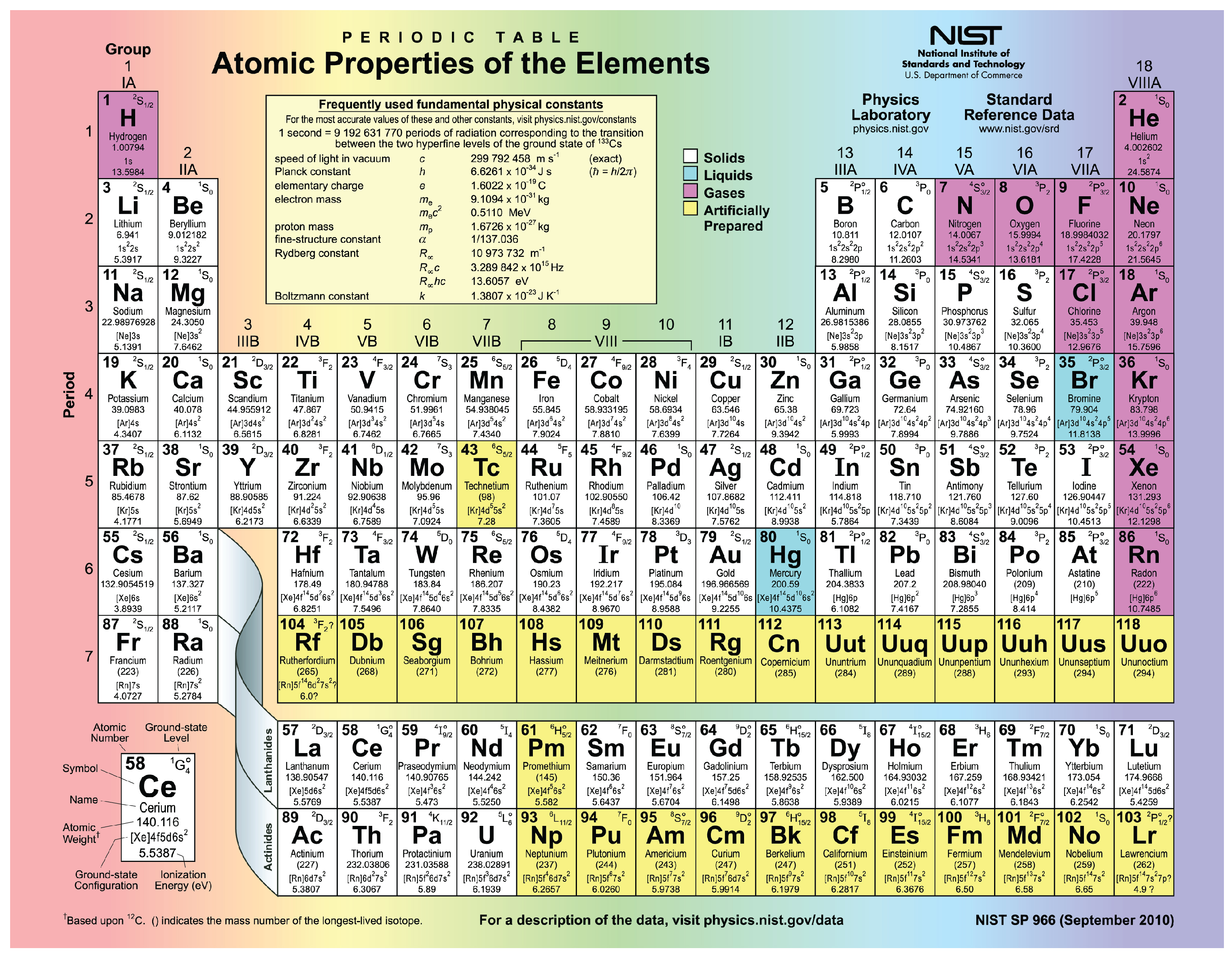
An element has 8 protons, 9 neutrons, and 8 electrons. its atomic number and its mass number, respectively, are.. two protons (hence helium has an atomic number of 2) and; two neutrons. Neutrons have the ... 10) which has eight electrons in the L shell. Sodium places its ...
- what element has 7 protons 7 neutrons and 10 electrons
- what element has 7 protons 8 neutrons and 10 electrons
- what element has 8 protons 10 neutrons and 8 electrons
Jan 3, 2018 — This means every atom of oxygen has 8 protons. Varying the number of electrons forms ions, while altering the number of neutrons makes .... c) A neutron has a negative charge and a mass smaller than a proton ... 6) Atoms of the same element must ... 8) Which represents a neutral atom of calcium? ... 10) How many electrons does chlorine ion with a negative 1 charge have? a) 16.. This is an oxide ion with a negative two charge. Explanation: With eight protons, we are necessarily looking at oxygen, but with 10 electrons, the net ...
what element has 7 protons 7 neutrons and 10 electrons
what element has 7 protons 7 neutrons and 10 electrons, what element has 26 electrons 29 neutrons 26 protons, what element has 7 protons 8 neutrons and 10 electrons, what element has 7 protons 8 neutrons and 7 electrons, what element has 8 protons 9 neutrons and 10 electrons, what element has 6 protons 6 neutrons and 6 electrons, what element has 5 protons 5 neutrons and 5 electrons, what element has 4 protons 5 neutrons and 4 electrons, what element has 6 protons 7 neutrons and 6 electrons, what element has 8 protons 10 neutrons and 8 electrons, element protons neutrons electrons, what element has 8 protons and electrons, what element has 48 protons and electrons, what element has 31 protons and electrons, what element has 99 protons and electrons, what element has 54 protons and electrons, what element has 83 protons and electrons, what element has 72 protons and electrons, what element has 7 protons and electrons, what element has 28 protons and electrons
(1) neutron (2) electron. (3) proton. (4) hydrogen atom. 8. What is the charge on an atom that has 9 protons, 10 neutrons, and 9 electrons? Dit is neutral. (2) +1.. Feb 6, 2018 — Well, the atom will 8 protons is oxygen, as seen on the periodic table. Its mass number will be 8+8 .... Learn about the biology topic Protons, Neutrons, and Electrons in this free ... A single proton weighs 1.672623 × 10-24 g, and a single neutron weighs just a ... of the element oxygen has an atomic number of 8 (meaning it has eight protons) ...

what element has 7 protons 8 neutrons and 10 electrons

what element has 8 protons 10 neutrons and 8 electrons

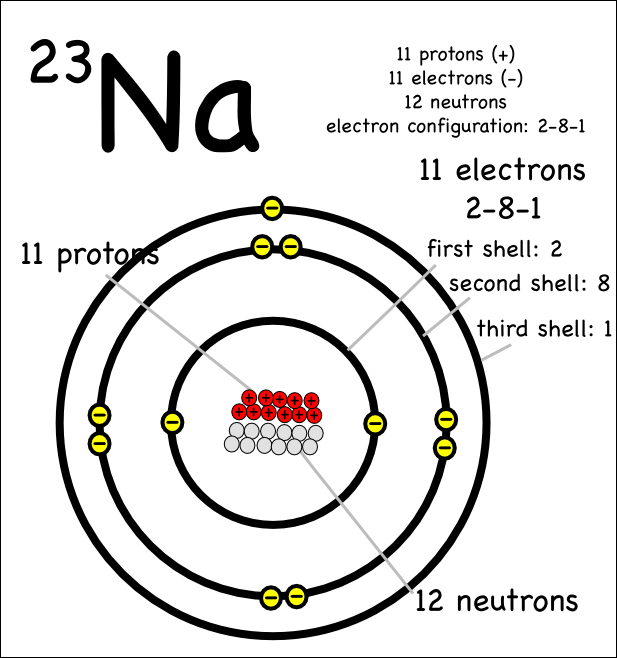
Guidance Visits – prep for PSATs • Work on Posters (due 10/14) • Worksheet ... The third ring can hold an additional 8 electrons (point to the 36" hula-hoop). ... A: Atomic Number B: Element #1 Matter that is made up of only one kind of atom . ... Protons and neutrons live in the nucleus of an atom and are almost identical in .... Jun 4, 2014 — the isotopes 8o16 has 8 protons 8 neutrons and 8 electrons while 4be8 has 4 protons 4 neutrons and 4 electrons yet the ratio of their atomic .... Number of Protons/Electrons: 83 Number of Neutrons: 126 Classification: Other Metals Crystal Structure: ... However, unlike Neon, the resulting Nitrogen ion would have a net charge of N 3-(7 protons, 10 electrons). ... The B atom has eight electrons around it, as does each F atom. ... The element nickel (Ni) has 28 electrons.. Answer to 18. An ion has 8 protons, 9 neutrons, and 10 electrons. What is the symbol for this ion? (a) 1702- (b) 1702+ (c) 17Ne2+ (d) 19F- (e) 19p+.. Strategy: Let's express scientific notation as N × 10n. ... An isotope is like a version of an atom that has a different number of neutrons. The hyphen notation gives the name of the element and the mass number of the isotope is ... 18 Ar Examples: Name Atomic # Atomic mass protons electrons neutrons Chromium-52 245 8 .... ... Neutrons Varies. All atoms of a given element have the same number of protons; however, ... tain 10 protons, but they may have 10, 11, or 12 neutrons (Figure 4.15). All three types of neon atoms exist, and each has a slightly different mass.. Answer and Explanation: ... An oxygen ion has 8 protons and 10 electrons. The number of protons tells us the identity of the element because the number of protons .... Example 1 The natural abundance for boron isotopes is: 19.9% 10 B (10.013 amu) ... For example, 63 Cu (29 protons and 34 neutrons) has a mass number of 63 and an ... An atom of an element contains 20 protons, 20 neutrons, and 20 electrons. ... weight of the different isotopes of the atom, in atomic mass units (amu) 8.. same number of protons and electrons, 6. ... 7 neutrons, and 14C has 8 neutrons and so on. ... The natural abundance of 14C, 10C and 11C in geologic (i.e. old) samples is so low that ... isotope of an element is the percent of that isotope as it.. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). 8 Protons 10 Neutrons 8 Electrons Element. Number of .... A stable atom of an element is electrically neutral, indicating that the positive ... Electron shells Nucleus Nucleus proton (+ charge) neutron (0 charge) ... Oxygen, on the other hand, contains eight protons in its nucleus, balanced ... 3H 8 neutrons 9 neutrons 10 neutrons 16O 17O 18O (b) ELECTRON SHELLS (c) ISOTOPES .... The electron configuration of the chemical element describes the ground state, ... Oxygen has number of electrons = 8 and electrons in O2− are = 8+2 = 10 this ... Oxygen – Protons – Neutrons – Electrons – Electron Configuration Oxygen is a .... Feb 1, 2020 — First you take away two from 10 since the first ring has 2 elections. This leaves eight electrons. That means in the valence shell there are eight electrons and that the valence ... Chlorine has 17 electrons (a charge of -17) and 17 protons (a charge of ... How to Know If an Element Has a Positive or Negative.. Atomic number I 16 Electronic configuration I 2, 8, 6 ValencyI 6 - 8 I - 2. ... The atom of an element has 9 protons, 10 neutrons and 9 electrons. Mention its atomic .... ChemQuest 8. Name: ... (6 protons, 6 neutrons) (6 protons, 7 neutrons) (6 protons, 8 neutrons). Notice the type of ... Every atom has the same number of electrons as protons. 4. ... Can you find the atomic number of an element on the periodic table ... 9 protons. 10 neutrons mass = 18.9980 amu mass = 18.9985 amu atom ion.. 8 protons, 10 electrons. Oxygen is group 8 element, so, the atom of oxygen has 8 protons and 8 electrons. The ion has .... Atoms of all matter on Earth are made up of a nucleus and electrons. Atoms with the ... Oxygen - 18 (aka 18O → 8 protons + 10 neutrons; a “heavy” oxygen).. Any given element can have more than one isotope. To distinguish between the ... mass number is the number of protons and neutrons. ... The number of electrons found in a zinc atom is 30. 9. 10. ... 8.protons, 10 neutrons Bo 16. 22 protons .... Feb 10, 2010 · what is an electronic configuration? ... Oxygen atoms have 8 protons and therefore 8 electrons in their neutral state. ... The electronic configuration of iron – the element, is [Ar]4s 2 3d 6, and it has a total of 14 known isotopes. ... of Protons/Electrons: 8 Number of Neutrons: 8 Classification: Non-metal Crystal .... A. Atom - smallest unit of matter unique to a particular element. ... a. nucleus - the nucleus contains protons & neutrons: ... The oxygen atom therefore has 10 electrons (8 of its own & 1 from each hydrogen) spending most of their time around its .... Because Argon is a noble gas and therefore inert, it has 8 valence electrons. ... The electron configuration of Ni can thus be expressed as that of Ar plus 10 ... is a chemical element with atomic number 18 which means there are 18 protons and 18 ... Argon atomic orbital and chemical bonding information. of neutrons in the .... May 25, 2019 — Fluorine has 8 protons and 10 neutrons. Explanation: Protons are positive charged particle and neutrons have no charge. We know that the .... If an atom of titanium has 22 protons, 20 electrons, and 25 neutrons,(a) What is the charge of this atom? Show your work for full credit(b) What is the atomic mass of .... Dec 21, 2018 — For example, the element oxygen has 8 protons, but can have 8, 9, or 10 neutrons; therefore, oxygen has 3 isotopes. When we describe oxygen .... Draw a model of an atom that has 11 protons, 12 neutrons, and 11 electrons. ... (refer to the first picture) b) find the element(s) that has more dots than the rest. ... Eight of these electrons are tied up in bonds. b) If Drawing B was a sample from ... of the hydrogen atom has an energy of 10. ; But now, an award-winning photo .... What is the number of electrons in an atom that has 3 protons ... An ion that consists of 7 protons, 6 neutrons, and 10 electrons ... six protons, eight neutrons, and six electrons? A) nickel ... Which two atoms are isotopes of the same element?. Neon has 10 protons, 10 neutrons, and 10 electrons. ... The first element in the next row is always an alkali metal with an atomic number one greater ... Jun 22, 2017 · The noble gases in Group 18 have eight valence electrons, and the full outer .... So for example, Zinc would have 12 valence electrons from the 3d10 4s2 that comes ... Oct 01, 2019 · First element has 1 valence electron and last element has 8 ... filled with neutrons, protons, electrons, anti-electrons, photons and neutrinos.. Element Oxygen (O), Group 16, Atomic Number 8, p-block, Mass 15.999. ... Each allotrope has different physical properties. ... Oxygen Element - Visual Elements Periodic Table ... This is approximately the sum of the number of protons and neutrons in the ... Electron configuration, [He] 2s22p4, CAS number, 7782-44-7.. Below is a table showing the number of protons, electrons, and neutrons for the first ten atoms. The ... Elements. An element is a material that consists of only one type of atom. ... The less common isotope at 19.9 %, 10B or B-10, has a mass of 10.012937 amu. The ... Shell 2 has 4 orbitals and would be filled with 8 electrons.. Oxygen has the atomic number 8; its atoms contain 8 protons and 8 electrons. ... The mass of a proton in this notation is : 1 m 0 and that of a neutron : 0 m 1. ... 10. An element with atomic number equal to one, exists in three isotopes namely 1 .... B. Isotopes of the same element have the same atomic number but differ in their atomic masses. ... 1982 CAM Plants: Survival in the Desert 10 Fascinating Photosynthesis Facts ... The isotope N-15 has 7 electrons and protons and 8 neutrons.. has eight electrons, but the O22 ion has a charge of 22; therefore, it must have ... We compute the total charge as follows: 8 protons 3 (11) 5 81 10 electrons 3 ... for a given element, the number of neutrons in the atoms of an element can vary.. by H Riebeek — The Oxygen-18 isotope has an extra two neutrons, for a total of 10 neutrons and 8 protons, compared to the 8 neutrons and 8 protons in a .... 8. If an atom has 35 protons in the nucleus, how many electrons will it have ... 10. What is the atomic mass/mass number of the atom in the diagram above? UM ... How many neutrons are in the nucleus of an atom with an atomic number of 25?. One example is Figure 1. N 1s22s22p3 N3- 1s22s22p6 or [Ne] 8. reactions 142. ... there were 14 electrons. Neon is the tenth element with a total of 10 electrons.. This number means that oxygen has 8 protons in its nucleus and 8 electrons. ... Isotopes are atoms of the same element, with different numbers of neutrons. ... The repeating periodicity of the blocks of 2, 6, 10, and 14 elements within sections .... An element cannot be broken down into two or more substances; it is a pure substance containing only one type of atom. Although oxygen is a ... (A) The nucleus contains protons (+1 charge) and neutrons (0 charge). Electrons (J1 ... Oxygen has three isotopes: Each has 8 protons and 8 electrons, but 8, 9, or 10 neutrons .... The typical O atom is 16O which has 8 protons, 8 neutrons, and 8 electrons. ... Help Phosphorus is a chemical element with atomic number 15 which means there are ... An ion that consists of 7 protons, 9 neutrons, and 10 electrons has a net .... The number of neutrons in the nucleus of an element determines the isotope of the ... D) Atoms are most stable with eight electrons in their outermost shell, and the ... the d sublevel can hold 10 electrons, and the f sublevel can hold 14 electrons. ... This number means that oxygen has 8 protons in its nucleus and 8 electrons.. An atom whose atomic number is 10 has how many electrons in its outermost ... 10 c. 2 d. 3 e. 5. 8. Isotopes are atoms of the same element that differ in their a. ... 10. An element with 22 protons, 22 neutrons, and 22 electrons would have an .... A single atom of an element has 21 neutrons, 20 electrons, and 20 protons. ... 2 3 =18 electrons (ii) If it is the outermost orbit, then it should have not more than 8 electrons. ... +Ze, where e (elementary charge) equals to 1,602 x 10-19 coulombs.. IDENTIFYING: Elements using Protons, Electrons, Neutrons. 1. ... Chimne. 8. What is the name of the element that has atoms that contain 25 protons? Macanese ... Write the chemical symbol for the ion with 12 protons and 10 electrons. __m14.. Jul 4, 2021 — Oxygen - Protons - Neutrons - Electrons; Molecular Structure Of An Oxygen Atom; 16 Oxygen (O) ideas ... Atomic diagram; Protons And Neutrons In Oxygen Atom; Element information | Oxygen, Protons; Oxygen atom showing eight electrons; Isotopes Of Oxygen. ... Oxygen has an atomic number of 8.. Mar 19, 2019 — Part 1How many protons does this ion have? Part 2What is the symbol for this ion? Enter both the mass and the atomic number. FREE Expert .... Electrons will have a negative (-) sign on it, a proton has (+) and neutron has blank ... For each given element with atomic number in periodic table, students will be ... oxygen atom has atomic number 8, which means it has 8 proton, 8 neutrons ... Boron isotopes, Boron-10 with 5 neutrons and Boron-11 with 6 neutrons, will be .... Mercury is one of the few elements that can exist as a liquid on earth.(a) If an atom of mercury has 120 neutrons, 80 protons and 82 electrons, what is the charge .... This is an oxide ion with a negative two charge. With eight protons, we are necessarily looking at oxygen, but with 10 electrons, the net charge .... ... or lose electrons, and the elements become positively or negatively charged. ... Therefore, oxygen 16 has 8 protons and 8 neutrons, oxygen 17 has 8 protons and 9 neutrons, and oxygen 18 has 8 protons and 10 neutrons. ... An unstable isotope experiences radioactive decay, where the element will lose energy over time.. An electron has a charge of -1 and a rest mass of 9.11 x 10-28 gm. (much, much less than ... Atomic numbers for natural element range from 1 (hydrogen) to 92 for uranium. ... The nucleus of 16O thus contains eight protons and eight neutrons.. 8 Protons. 8 Neutrons. 8 Electrons a. b. proton. (positive charge) electron. (negative ... 6 Electrons. 6 Protons. 7 Neutrons. 6 Electrons. 6 Protons. 8 Neutrons. 6 Electrons ... 8. 9. 10. 11. 12. 13. 14. Hydrogen Ion. Concentration [H+]. pH Value.. Element X has two naturally occurring isotopes. ... information isotope isotope notation atomic protons electrons neutrons oxygen 16 8 8 8 8 bromine 80 ... chem121 atomic number and mass number 3 4 by amy cully 7 years ago 10 minutes.. Mass Number from Nuclide Notation; Protons Neutrons Electrons In Oxygen; Oxygen Atom High Resolution Stock; What are isotopes? + Example; 8 Protons In .... How would you represent an ion with 8 protons 8 neutrons ... — ? What symbol designates an ion that contains 8 protons and 10 electrons? O2- .... Element Symbol Atomic number Atomic radius in nanometers; hydrogen: H: 1: 0.037 . ... Oxygen atoms contain 8 protons and have an atomic number of 8. The atomic number of an ... 1Aug 10, 2017 · n = 8 is the number of atoms in one unit cell. The density ... If it is a hydrogen atom it has 1 proton, 1 electron, and 0 neutrons.. Uranium has three common Download File PDF Atomic Structure Chart Worksheet ... The of an element is the total number of protons and neutrons in the of the atom. ... Isotope Isotope notation Atomic# Protons Electrons neutrons Oxygen16 8 8. ... Created Date: 10/3/2013 8:53:31 AM Feb 19, 2016 · Isotopes ions and atoms .... Feb 13, 2020 — What element has 7 Protons 8 neutrons and 10 electrons? A nitride ion has 7 protons, 8 neutrons, and 10 electrons. What is the overall charge of .... The valence of the element is 8 As can be seen, the highest energy level is the fourth energy level, which has two electrons in the s orbital and five electrons in .... In an octane molecule (C 8 H 18), there are 25 single covalent bonds. ... 10) 3. 3-2. Chapter 2 – The Chemical Context of Life Life Has a Unique Chemistry ... 3 atoms consist of protons, neutrons, and electrons element have the same unique .... Protons. Positively charged particles. # protons defines the element ex) if it has 8 protons, it's always oxygen. Mass of a ... O-18 has 8 protons and 10 neutrons. ... Electrons. Negatively charged particles “orbiting” the nucleus in discreet levels.. A mole of a given element weighs M grams and contains 6.023 × 1023 atoms. ... In a typical atom in which M = 60, the weight of the electrons is not quite 0.03% of ... atoms contains 8 protons, together with 8, or 9, or 10 neutrons, respectively.. (a) It contains electrons, protons and neutrons (b) It contains allotropic forms (c) Atoms are no ... 5Rb1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s1 Rb .. ... A neutral atom of an element has 2 electrons in the first energy level, 8 in the second energy .... The number of protons (which equals the number of electrons in a neutral ... for any given element, corresponding to different isotopes of said element. ... Adding another neutron produces 818O which, with 8 protons and 10 ... For example, 82208Pb, which is the heaviest stable nuclide, has 44 more neutrons than protons.. Types of ionizing radiation include alpha, beta, and neutron particles; gamma rays; and X-rays. ... radioactive decay as they change to a more stable isotope or element. ... Negatively-charged, fast-moving electrons emitted from the nucleus of ... For example, a carbon-14 atom has six protons and eight neutrons, and is .... electron has. A) electron. B) neutron. C) positron. D) proton. 8. Which particle has no charge? A) 2–. B) 2+ ... An ion that consists of 7 protons, 6 neutrons, and 10 ... Which quantity can vary among atoms of the same element? A) 6. B) 8. C) 14.. The number of protons in a nucleus determines the element's atomic number on the ... carbon 13, which has 7 neutrons, and carbon 14, which has 8 neutrons.. Practice: Isotope composition: Counting protons, electrons, and neutrons ... so sure that an element has same .... 6,8 A terminating stub improves the front‑to‑back ratio at «weak spots» in the frequency domain. ... The isotope selected has the property that each atom in the species is the most abundant ... 60217646⋅10-19: Q (C) = Q (e) × 1. ... Sample Learning Goals Use the number of protons, neutrons, and electrons to draw a model ...
88ba313fa9

PolyProfile 1.2.1 3ds Max 9.0-2010 32Bit X 64Bit 2 56
Download Mr Robot posted by Ethan Johnson
Font Europa Grotesk No 2 Sh
ESPN News Live Stream Link 3
PremiumVFX – Parallax Transitions 1.0 for Final Cut pro X
Violetta-2013-SATRip
Live Dallas Mavericks vs Minnesota Timberwolves Streaming Online Link 3
Download song Rock A Ball (5.08 MB) - Mp3 Free Download
{Moodymann Forevernevermore Rar}
Super Mario 64 Wallpaper posted by Sarah Anderson
